

Resources & Support
HomeResources & SupportDrug Target Insights
MingCelerDrug Target Insights
-
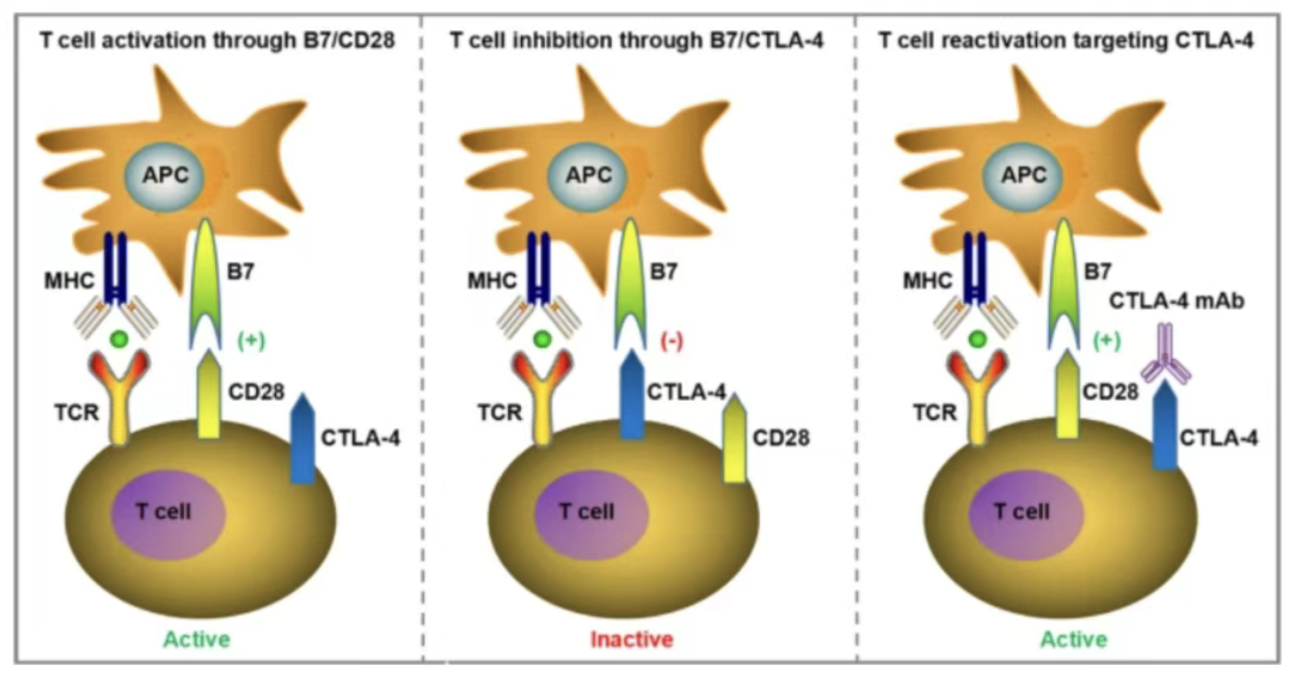 Apr 21, 2026Drug Target Outlook | CTLA-4: The Core Immune Checkpoint, a Hub Target for Dual Regulation in Cancer and AutoimmunityCTLA-4 (CD152) holds the distinction of being the first immune checkpoint inhibitor pathway sanctioned by the FDA for oncology applications. Following its discovery in the 1990s, the development of Ipilimumab (Yervoy®) established CTLA-4 as a critical target in tumor immune evasion. Currently, CTLA-4 targeted therapies are a vital component in the clinical management of refractory solid tumors, including melanoma and renal cancer.
Apr 21, 2026Drug Target Outlook | CTLA-4: The Core Immune Checkpoint, a Hub Target for Dual Regulation in Cancer and AutoimmunityCTLA-4 (CD152) holds the distinction of being the first immune checkpoint inhibitor pathway sanctioned by the FDA for oncology applications. Following its discovery in the 1990s, the development of Ipilimumab (Yervoy®) established CTLA-4 as a critical target in tumor immune evasion. Currently, CTLA-4 targeted therapies are a vital component in the clinical management of refractory solid tumors, including melanoma and renal cancer. -
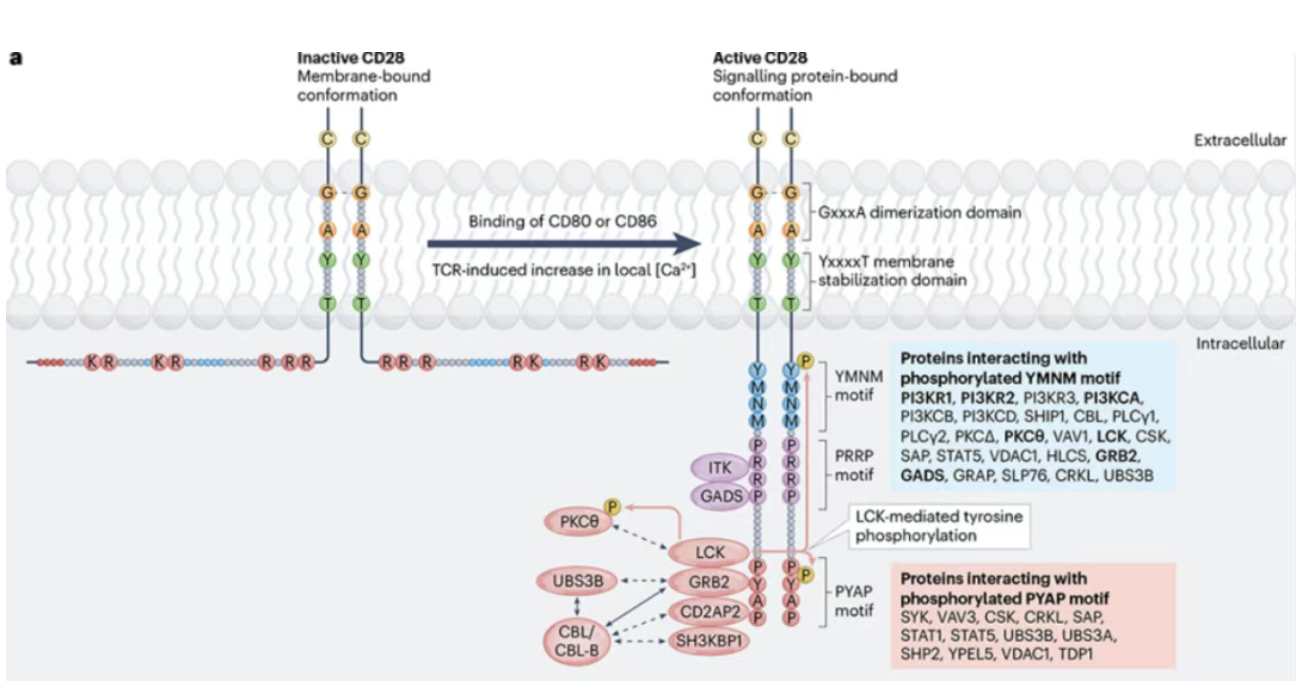 Apr 15, 2026CD28: The Core Co-stimulatory Molecule in Adaptive Immunity and AutoimmunityIn the adaptive immune response, T cell functional activation relies on a critical two-signal regulatory mechanism. T cell receptor (TCR) recognition of the MHC-antigen peptide complex on the surface of antigen-presenting cells (APCs) provides the first signal. The co-stimulatory receptor CD28 binding to its ligands B7.1 (CD80) and B7.2 (CD86) on APCs provides the essential second signal. Together, these signals drive full T cell activation, clonal proliferation, and effector differentiation. The CD28 signal drives T cell clonal expansion, effector differentiation (e.g., IL-2 secretion), and metabolic reprogramming. Its precise regulation has become a central target in tumor immunotherapy and autoimmune disease intervention.
Apr 15, 2026CD28: The Core Co-stimulatory Molecule in Adaptive Immunity and AutoimmunityIn the adaptive immune response, T cell functional activation relies on a critical two-signal regulatory mechanism. T cell receptor (TCR) recognition of the MHC-antigen peptide complex on the surface of antigen-presenting cells (APCs) provides the first signal. The co-stimulatory receptor CD28 binding to its ligands B7.1 (CD80) and B7.2 (CD86) on APCs provides the essential second signal. Together, these signals drive full T cell activation, clonal proliferation, and effector differentiation. The CD28 signal drives T cell clonal expansion, effector differentiation (e.g., IL-2 secretion), and metabolic reprogramming. Its precise regulation has become a central target in tumor immunotherapy and autoimmune disease intervention. -
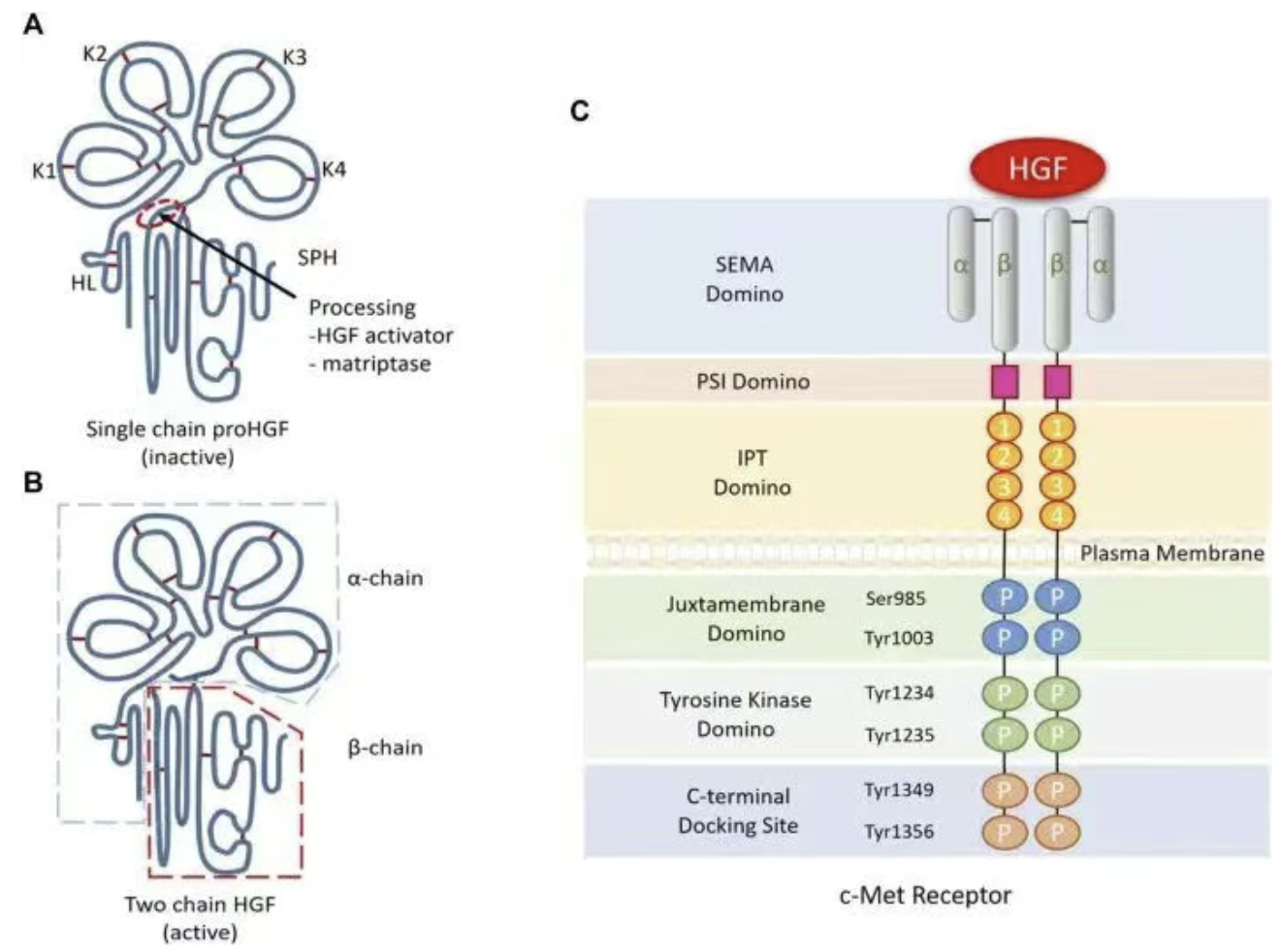 Apr 08, 2026The HGF/MET Signaling Axis: Why It Matters in CancerCancer does not grow and spread on its own. It depends on signals that tell tumor cells to survive, divide, move, and resist treatment. One of the most important of these signaling systems is the HGF/MET axis.
Apr 08, 2026The HGF/MET Signaling Axis: Why It Matters in CancerCancer does not grow and spread on its own. It depends on signals that tell tumor cells to survive, divide, move, and resist treatment. One of the most important of these signaling systems is the HGF/MET axis. -
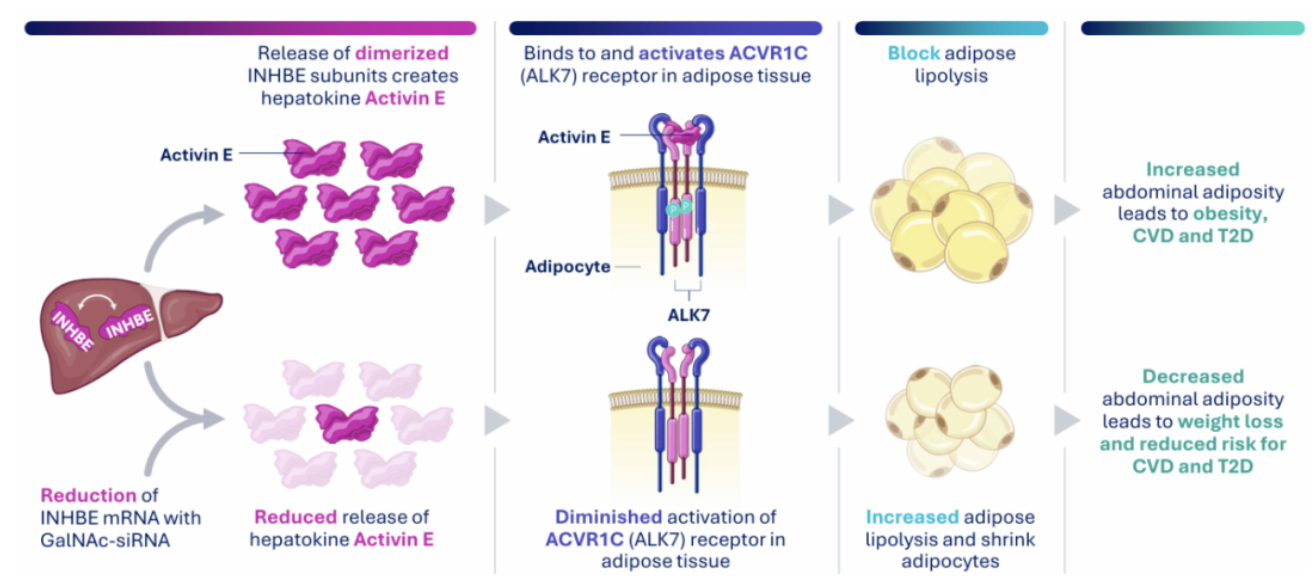 Apr 07, 2026INHBE: From a Silent Gene to a Hepatic Hub Target in the Fight Against Metabolic DiseasesThe INHBE gene, officially named Inhibin Subunit Beta E, is located on human chromosome 12q13.3 and encodes a secreted protein belonging to the TGF-β superfamily. Traditionally, due to its sequence homology with the inhibin/activin family, it was long speculated to play a role in reproduction and development. However, several recent landmark genetic studies have rewritten its story, redefining it as a core target in the field of metabolic regulation.
Apr 07, 2026INHBE: From a Silent Gene to a Hepatic Hub Target in the Fight Against Metabolic DiseasesThe INHBE gene, officially named Inhibin Subunit Beta E, is located on human chromosome 12q13.3 and encodes a secreted protein belonging to the TGF-β superfamily. Traditionally, due to its sequence homology with the inhibin/activin family, it was long speculated to play a role in reproduction and development. However, several recent landmark genetic studies have rewritten its story, redefining it as a core target in the field of metabolic regulation. -
 Apr 03, 2026Complement Factor B and Humanized Animal Models: Interpreting Targeted Therapy Research for Kidney DiseasesIn kidney diseases caused by abnormal complement system activation, the overactivation and amplification of chain reactions in the alternative complement pathway are the core causes of renal damage. This leads to the accumulation of a protein fragment called C3 in the glomerulus, activating the final damage pathway of the complement system and progressively destroying kidney function. Complement Factor B (abbreviated as CFB) is a key "driver" in this "pathogenic chain" and an indispensable core molecule in the alternative complement pathway. Its main role is to assist in assembling two key "tools" (C3 convertase and C5 convertase), maintaining the "positive feedback loop" of this abnormal reaction and sustaining the damage [1].
Apr 03, 2026Complement Factor B and Humanized Animal Models: Interpreting Targeted Therapy Research for Kidney DiseasesIn kidney diseases caused by abnormal complement system activation, the overactivation and amplification of chain reactions in the alternative complement pathway are the core causes of renal damage. This leads to the accumulation of a protein fragment called C3 in the glomerulus, activating the final damage pathway of the complement system and progressively destroying kidney function. Complement Factor B (abbreviated as CFB) is a key "driver" in this "pathogenic chain" and an indispensable core molecule in the alternative complement pathway. Its main role is to assist in assembling two key "tools" (C3 convertase and C5 convertase), maintaining the "positive feedback loop" of this abnormal reaction and sustaining the damage [1].

 Login
Login CN
CN

